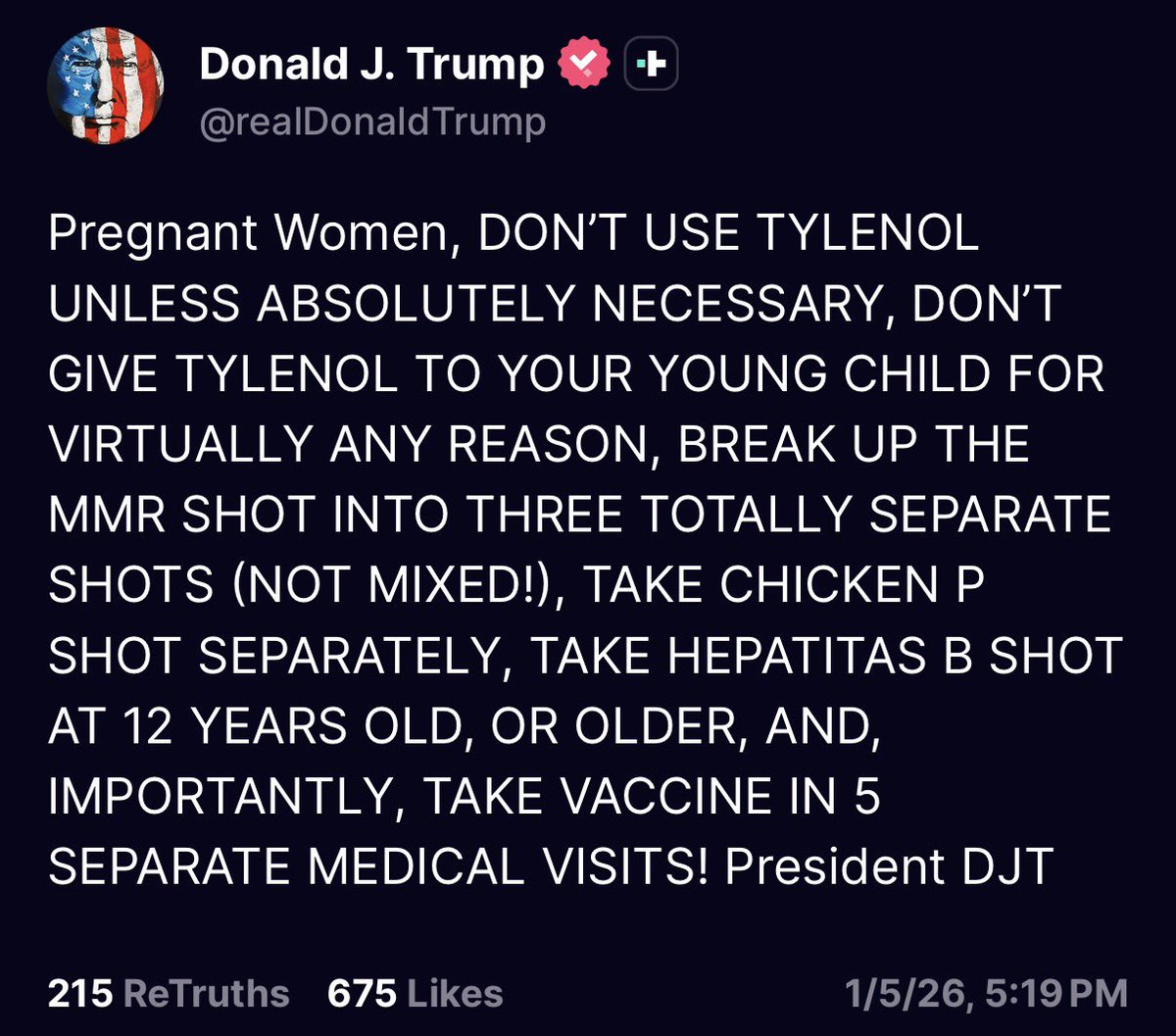
President's Health Guidance Sparks Debate Across Public Health and Medical Communities
In a surprising turn of public health guidance, statements attributed to President Donald J. Trump regarding medication use and childhood vaccinations have drawn widespread attention and prompted rapid responses from medical professionals, policymakers, and the public. The guidance, which emphasizes caution around common medications and advocates for altering established vaccination schedules, raises questions about medical ethics, regulatory oversight, and the practical implications for families navigating vaccination decisions.
Historical Context and Evolution of Vaccination Policy
Vaccination policies have long been shaped by a balance between protecting individual health and achieving herd immunity to safeguard communities. Over decades, the United States has built a comprehensive framework for routine childhood vaccines, guided by recommendations from the Advisory Committee on Immunization Practices (ACIP) and coordinated with national distribution systems by health agencies. The standard schedules, including combined vaccines such as the MMR (measles, mumps, rubella) and the varicella (chickenpox) vaccine, have been developed through extensive clinical trials and post-marketing surveillance to maximize protection while minimizing adverse events.
Public health officials emphasize that vaccines are among the most effective tools for preventing serious infectious diseases. Alterations to recommended schedules are typically guided by evidence-based risk assessments and are discussed extensively within medical communities before any public dissemination. Historical episodes, such as smallpox eradication campaigns, polio vaccination drives, and recent measles outbreaks, illustrate how shifts in public health guidance can influence disease dynamics, healthcare costs, and societal trust.
Medication use in pregnancy and early childhood is another area of careful scrutiny. Acetaminophen, commonly known by the brand Tylenol, has long been used to manage fever and discomfort during pregnancy and in children, with clinical guidelines prioritizing the minimum effective dose and duration. Routine medical advice generally emphasizes caution but acknowledges that, in many cases, the benefits of symptom relief outweigh potential risks. Policy discussions around analgesic use during pregnancy typically revolve around evidence-based risk assessments, pharmacovigilance data, and patient-centered decision-making.
Economic Impact and Healthcare System Considerations
If such guidance were to influence patient behavior significantly, several economic and operational effects could follow. First, altered vaccination schedules could affect immunization delivery systems, including pharmacy networks, pediatric practices, and school-entry vaccination compliance. Delays or fragmentation of vaccination could lead to increased susceptibility in populations, potentially elevating the risk of outbreaks and associated healthcare expenditures, including hospitalizations, emergency room visits, and intensified public health campaigns.
From a cost perspective, routine childhood vaccines represent substantial long-term savings by reducing the incidence of preventable diseases. Shifts toward non-standard dosing regimens or delayed vaccination could undermine these savings and introduce higher short-term costs for families and communities as disease prevention becomes more complex to manage. Pharmacy supply chains and vaccine manufacturers could face recalibration challenges if demand patterns deviate from established schedules.
In pregnancy-related medication guidance, changes in recommended practices can influence prenatal care models, clinician training, and patient education programs. Providers may need to allocate additional time for counseling on medication risks and alternatives, potentially impacting appointment availability and healthcare utilization. Insurers and payer systems would also assess coverage implications for any proposed changes in treatment protocols or precautionary measures.
Regional Comparisons and Global Perspectives
Across regions, vaccination policies and medical guidance vary based on local epidemiology, healthcare infrastructure, and cultural attitudes toward preventive care. Some countries prioritize broader vaccine schedules, while others emphasize targeted immunization strategies aligned with disease prevalence. Global health organizations advocate for maintaining high vaccination coverage and clear, evidence-based messaging to minimize confusion and ensure consistent protection against infectious diseases.
In areas with recent outbreaks or vulnerable populations, public health authorities may implement interim recommendations that differ from standard schedules while still aiming to preserve overall population immunity. Comparisons of adverse event reporting systems reveal how surveillance practices influence perceived safety and public confidence, underscoring the importance of transparent communication and rigorous post-marketing monitoring in maintaining trust.
Public Reaction, Communication, and Trust
Public reaction to any guidance that appears to alter foundational medical practices tends to be mixed, with varied interpretations shaped by personal experiences, media coverage, and trust in institutions. Supporters often emphasize precaution and patient autonomy, while critics caution against undermining proven prevention strategies. Social media dynamics can amplify both concerns and misinformation, making rapid, accurate communication essential for public health authorities and professional societies.
Health communication experts stress the value of clear explanations about risk, benefit, and uncertainty. For parents, the core questions typically revolve around the safety of medications during pregnancy and early childhood, the rationale for recommended vaccine schedules, and the availability of alternatives when specific concerns arise. Clinicians play a central role in conveying personalized guidance, helping families navigate complex choices while maintaining adherence to evidence-based practices.
Clinical, Ethical, and Regulatory Considerations
From a clinical standpoint, any departure from established, evidence-based guidelines warrants rigorous review and transparency. Medical ethics emphasize patient autonomy paired with non-maleficence, requiring clinicians to present balanced information about potential harms and benefits. Regulatory bodies and professional associations would likely assess such guidance to determine whether it aligns with scientific consensus and whether it warrants update to clinical practice guidelines.
Moreover, the regulatory environment surrounding vaccines and medications involves post-market surveillance, pharmacovigilance, and ongoing research. Even if new recommendations emerge, they must be supported by robust data and subjected to peer review. Independent replication and long-term outcome assessments are critical to integrating any policy shift into standard care.
Implications for Public Health Planning
Public health planning hinges on predictable patterns of vaccination uptake and medication use, which in turn influence disease prevention strategies and resource allocation. Maintaining high vaccine coverage remains a central objective for health departments, hospitals, and community health programs. Any guidance suggesting a segmented or delayed vaccination approach would necessitate careful modeling to anticipate potential outbreak scenarios and to design targeted interventions in schools, clinics, and community centers.
Emergency preparedness and response frameworks also rely on consistent messaging. During infectious disease surges, coordinated communication helps minimize confusion and ensure that individuals receive timely protections. Public health agencies might need to issue clarifications or reaffirm adherence to evidence-based vaccination schedules to safeguard community health.
Historical Lessons for Future Guidance
Historical patterns in public health underscore the importance of consistency, transparency, and scientific rigor when issuing guidance that affects large populations. Clear separation between policy decisions, clinical recommendations, and political messaging helps preserve public trust and reduces the risk of misinterpretation. When new data emerge, the responsible course is to publish findings, subject them to review, and update guidelines in a manner that stakeholders can understand and implement.
As researchers continue to study the safety profiles of common medications in pregnancy and early childhood, it remains crucial to distinguish between preliminary findings and established conclusions. Policymakers and health professionals can then translate evolving evidence into practical recommendations that protect health while respecting individual circumstances.
Conclusion: Navigating Uncertainty with Evidence-Based Practice
The dialogue surrounding medication use during pregnancy and childhood vaccination schedules highlights the dynamic interplay between science, policy, and public perception. While vigilance in medical decision-making is essential, maintaining confidence in proven preventive measures is equally important. The goal for health systems is to provide clear, consistent guidance grounded in the best available evidence, complemented by robust surveillance and open channels for public inquiry.
In the coming weeks and months, stakeholders across medicine, public health, and civic life will assess the real-world impact of any new guidance. Communities will watch for data from ongoing studies, watchful for trends in disease incidence, vaccination compliance, and overall health outcomes. The ultimate measure of success will be the continued protection of vulnerable populations, reductions in preventable illness, and sustained trust in medical recommendations that are transparent, rigorous, and responsive to new information.