Synthetic Psilocybin Shows Breakthrough Promise for Treatment-Resistant Depression in Late-Stage Trials
Major Step Forward in Mental Health Treatment
A synthetic form of psilocybin, the psychoactive compound found in so-called "magic mushrooms," has demonstrated significant efficacy in two late-stage clinical trials for patients suffering from treatment-resistant depression. The investigational drug, known as COMP360, is being developed by Compass Pathways, a U.K.-based biotechnology company specializing in mental health innovation.
On February 17, Compass Pathways announced that COMP360 achieved statistically significant improvements in depressive symptoms among participants who had previously failed to respond to standard antidepressant therapies. The results, marking a major milestone in psychiatric medicine, have set the stage for possible regulatory approval as early as 2027. If approved, COMP360 would become the first fully licensed psychedelic-based treatment for depression.
Details of the Breakthrough Trials
The pivotal studies evaluated COMP360 in hundreds of participants diagnosed with treatment-resistant depression (TRD)—a form of major depressive disorder that does not improve after two or more conventional therapies.
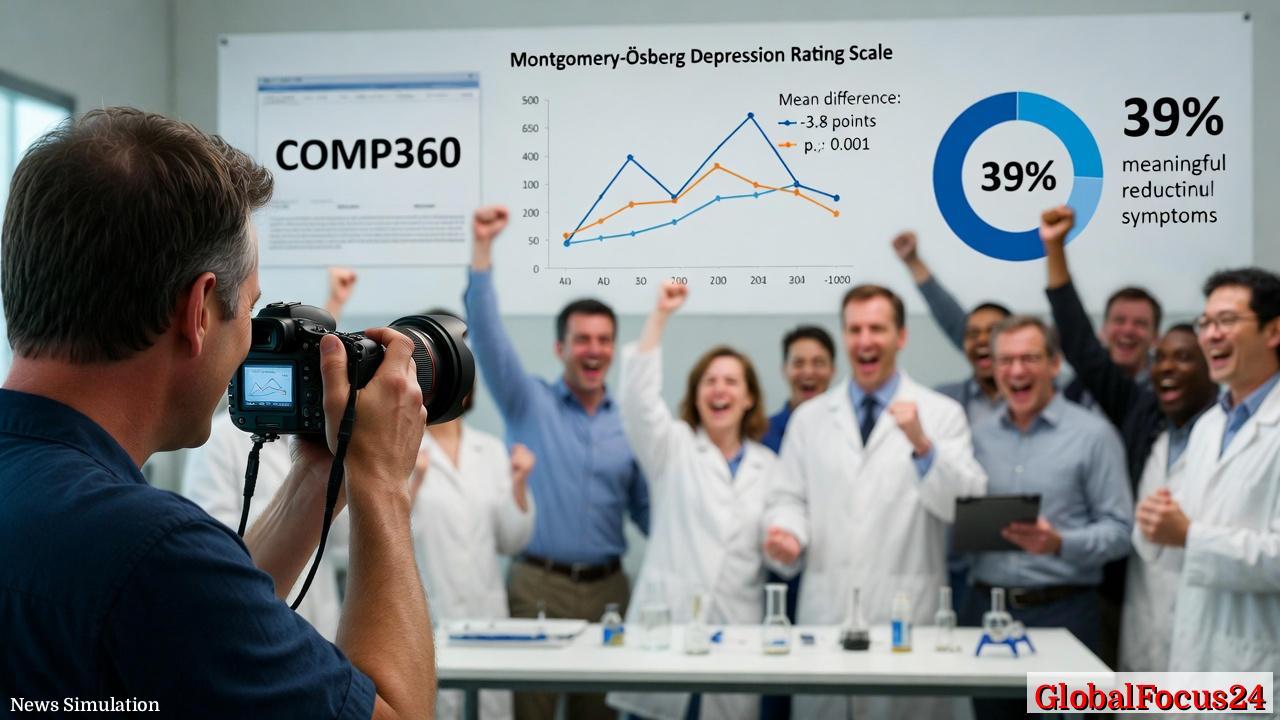
In the first trial, two administrations of a 25 mg dose produced a highly significant reduction in depression severity, as measured by the Montgomery-Åsberg Depression Rating Scale (MADRS), compared with a 1 mg control dose. The difference in mean scores at week six averaged -3.8 points, a result that exceeded the trial’s primary efficacy target. Nearly 39% of participants receiving the 25 mg dose experienced a clinically meaningful improvement—defined as a reduction sufficient to alter daily functioning and quality of life.
A second late-stage study replicated these strong findings, confirming the drug’s short- and medium-term benefits. Researchers observed reductions in depressive symptoms as early as one day after administration, with many improvements sustained through the entire follow-up period. This rapid onset contrasts with traditional antidepressants, which often require weeks of consistent use before measurable effects appear.
Scientific Foundations and Pharmacological Approach
Psilocybin, a naturally occurring tryptamine compound, exerts its effect primarily through serotonin 5-HT2A receptor activation in the brain, leading to temporary changes in perception, mood, and cognitive flexibility. Scientists believe these brain-level changes can “reset” dysfunctional neural circuits associated with depression and anxiety.
COMP360 is a synthetic and pharmaceutical-grade formulation of psilocybin designed for standardization and predictable dosing—an essential factor for regulatory approval and medical use. The therapy is administered under careful psychological support, typically within a guided session lasting several hours with trained professionals observing and assisting patients throughout the experience.
This structured approach sets COMP360 apart from unsupervised or recreational psychedelic use. Clinical frameworks ensure both safety and therapeutic integration, helping participants process the often emotionally intense experience in a stabilizing environment.
Growing Demand for New Depression Treatments
Treatment-resistant depression remains one of the most challenging areas in psychiatry. According to the World Health Organization, more than 300 million people worldwide suffer from depression, and an estimated one-third of patients fail to respond adequately to existing antidepressant medications. For many, options beyond therapy and selective serotonin reuptake inhibitors (SSRIs) are limited.
The economic burden is equally immense. The U.S. Substance Abuse and Mental Health Services Administration has reported annual productivity losses exceeding $200 billion due to depression-related absenteeism and reduced work performance. A faster-acting and more durable treatment could dramatically reduce these social and financial costs while improving overall quality of life for millions.
How COMP360 Compares Regionally and Historically
Across the globe, researchers and regulators have been watching psilocybin’s progress closely. In recent years, several countries, including Canada and Australia, have permitted limited therapeutic use of psychedelic substances under strict medical supervision. Australia, for example, became the first nation to formally reclassify psilocybin as a controlled substance for clinical prescription in 2023, marking a cautious but historic shift in psychiatric practice.
In the United States, the Food and Drug Administration has already granted Breakthrough Therapy Designation to COMP360, recognizing its potential to offer substantial advantages over existing treatments. This status accelerates regulatory review and reflects growing confidence in the safety and efficacy data emerging from controlled trials.
Historically, psychedelic research was largely halted in the early 1970s following global scheduling bans. Only in the past two decades, through advances in neuroscience and imaging technology, has scientific interest in psychedelics experienced a resurgence. Today’s studies are far more controlled, data-driven, and ethically supervised than those of past decades, aligning with modern drug development standards.
Patient Experiences and Clinical Observations
While the mechanism of action continues to be explored, clinical staff have reported that many patients describe the COMP360 sessions as emotionally cathartic, allowing deep introspection and relief from entrenched feelings of despair. The combination of the psychedelic effect and ongoing psychological support may enable participants to develop new perspectives on previously overwhelming emotional patterns.
However, the therapy is not without challenges. The sessions can evoke strong emotional responses or temporary anxiety, which requires professional guidance to manage safely. Compass Pathways’ trials include rigorous safety screening and follow-up to mitigate risks of adverse psychological reactions. So far, the most common side effects have been mild—and transient—including headache, nausea, and brief episodes of anxiety.
Regulatory Outlook and Timeline for Approval
Both the U.S. Food and Drug Administration and the European Medicines Agency are reviewing data from the COMP360 Phase 3 trials. Given the depth of evidence and the strong safety profile, analysts suggest that approval decisions could arrive as early as 2027 in the United States, with Europe possibly following within a similar timeframe.
If authorized, COMP360 could become the first psychedelic compound to achieve full medical licensing status in over half a century, opening pathways for a new class of psychiatric treatments. The approval could also trigger a surge in pharmaceutical investment toward other psychedelic compounds for conditions such as post-traumatic stress disorder, anorexia nervosa, and generalized anxiety disorder.
Economic Projections and Industry Implications
The global depression treatment market, valued at over $15 billion in 2024, is projected to expand rapidly if psychedelic-based therapies are commercialized. Analysts anticipate that breakthrough approvals could double the market’s value within a decade.
Compass Pathways, already partnered with leading academic research centers, plans to expand its clinician training programs and infrastructure for certified psychedelic therapy sites. Ensuring accessibility will be a primary challenge. Psilocybin therapy requires in-person administration and intensive professional oversight, which may initially limit availability to specialized clinics. Nonetheless, the long-term commercial and therapeutic potential remains substantial.
Broader Public and Medical Reaction
Public response to the COMP360 results has been mixed but increasingly optimistic. Advocates for mental health innovation view the findings as validation of decades-long efforts to reexamine the therapeutic potential of controlled substances. Mental health charities and research coalitions have expressed hope that new biologically distinct treatments can fill critical gaps left by conventional antidepressants.
Still, some medical professionals urge cautious optimism. Long-term safety and durability of remission rates must be verified through extended follow-up studies. Critics emphasize that psychedelic experiences, while transformative for some, may not be universally beneficial or appropriate for every patient, particularly those with certain mental health risk factors.
The Next Chapter for Psychedelic Science
As global health systems continue searching for faster, more effective interventions against mental illness, synthetic psilocybin represents one of the most promising—and controversial—frontiers in pharmacology. The COMP360 trials have moved the conversation decisively from speculative exploration to actionable medical science.
If regulatory approval proceeds as expected, psilocybin-assisted therapy could redefine the treatment landscape for millions of people struggling with depression that conventional medicine has failed to address. The coming years will reveal whether this modern reimagining of a once-forbidden compound can deliver on its promise to reshape the future of mental healthcare.