New Research Reveals Skull Veins Can Reshape to Support Brain’s Immune Defense
Groundbreaking Discovery in Brain-Immune System Link
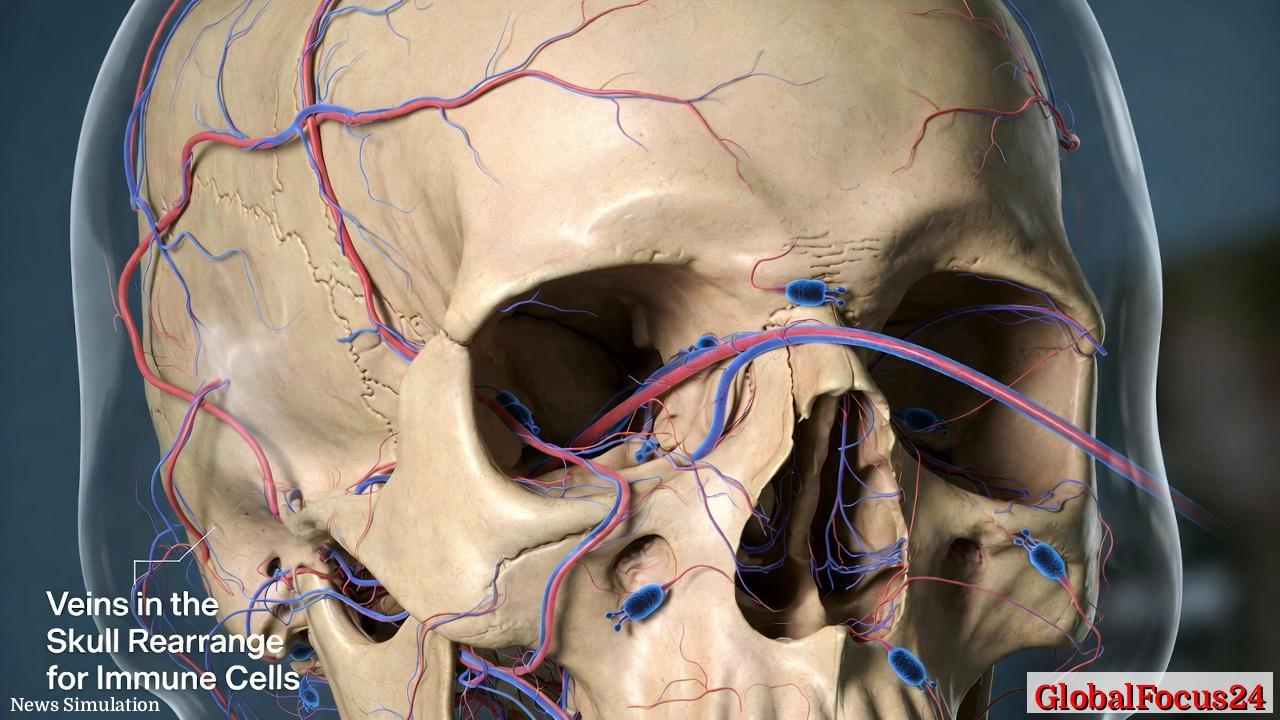
In a major advance in neuroscience and immunology, researchers have found that veins in the skull can actively reshape their borders to accommodate immune cells patrolling the brain’s protective layers. The discovery sheds new light on the dynamic relationship between the brain and the body’s immune system, suggesting that the skull plays a far more active role in managing inflammation and maintaining neural health than previously understood.
The finding redefines the long-held view that the skull’s vascular system is a rigid structure. Instead, scientists now know that certain skull veins possess the flexibility to rearrange their borders — effectively opening controlled passageways for specialized immune cells. Through these adaptive processes, the skull serves as a gatekeeper, helping to detect and respond to potential threats before they reach the delicate tissue of the brain.
The Brain’s Hidden Drainage Network
For decades, researchers believed that the brain was largely isolated from the immune system, protected by the blood-brain barrier. However, recent discoveries have transformed that understanding. Scientists have identified microscopic channels — informally called “skull drains” — that connect the bone marrow of the skull directly to the coverings of the brain. These drains allow immune cells to travel efficiently from the skull to the meninges, the protective membranes surrounding the brain.
This new research advances that concept further. It demonstrates that the venous structures intertwined with these channels are not static. Instead, they adapt their shape and boundaries, likely helping regulate the flow of immune cells during both normal immune surveillance and in response to injury or disease. By opening or tightening these microscopic gates, the skull appears to fine-tune immune access to the brain, acting almost like a border patrol system at the threshold of the central nervous system.
Evolutionary and Biological Significance
The evolutionary logic of this vascular adaptability is compelling. The brain, as the body’s most sensitive organ, requires tight control over immune access. Too few immune cells and pathogens might flourish unchecked. Too many or excessively aggressive immune responses, however, could trigger damaging inflammation.
The skull’s ability to rearrange its venous borders could therefore represent an elegant evolutionary compromise. It grants immune cells the access they need while maintaining the integrity of the brain’s environment. Similar adaptive processes — though far less sophisticated — can be observed in other parts of the body, such as the intestinal barrier or lymphatic vessels, where selective permeability ensures a balance between defense and stability.
This discovery positions the skull not only as a protective shell but as an intelligent gatekeeper that interacts constantly with the immune system. Its structural plasticity reveals how integrated the brain and body truly are, despite earlier theories that depicted them as separate entities.
Implications for Brain Injury and Disease
Beyond its biological intrigue, this research has far-reaching clinical implications. If skull veins and drains help regulate immune traffic to the brain, they may play key roles in conditions such as traumatic brain injury, stroke, and neurodegenerative disease.
In cases of head trauma, for example, the skull’s vascular flexibility might determine how quickly immune cells can mobilize to repair damaged tissue. Too much immune infiltration could worsen swelling and secondary injury; too little could delay recovery. Understanding how to modulate this venous responsiveness may lead to therapies that harness the body’s own immune regulation mechanisms for faster healing and reduced complications.
In neurodegenerative conditions like Alzheimer’s or multiple sclerosis, the picture may be even more complex. Researchers speculate that malfunctions in these skull drainage pathways could disrupt the clearance of waste products or immune signaling molecules, contributing to inflammation and neuronal decline. Future treatments might aim to restore or enhance the skull’s natural immune drainage process, potentially slowing progression of such disorders.
From “Immune Privilege” to Dynamic Communication
The study is part of a growing body of research overturning the once-dominant concept of “immune privilege,” which held that the brain was largely inaccessible to immune cells. Over the past decade, advanced imaging and mapping technologies have revealed a constant, finely tuned dialogue between the brain and immune system.
Flow cytometry and 3D microscopy techniques have allowed researchers to observe immune cells traveling through skull marrow cavities and crossing microvascular borders into the meninges. These interactions appear to increase with age, infection, or stress — situations that demand heightened immune surveillance.
By illustrating that skull veins themselves can remodel to accommodate this process, scientists are now adding a crucial new dimension to the picture. The brain is no longer viewed as merely insulated but as selectively connected, protected not by isolation but by intelligent control of access.
Global and Historical Context
Neuroscience has a long history of treating the skull as a static barrier. Early anatomical texts from the Renaissance described it primarily as bone armor protecting the brain’s soft tissue. Even in the 20th century, medical imaging and neurosurgery largely reinforced this mechanical view. It was only with the advent of molecular biology and high-resolution imaging in recent decades that the skull’s more subtle physiological roles began to emerge.
Comparatively, similar systems of skull-marrow communication have been explored in other species, including rodents and primates, where immune cell migration through skull channels has been documented. These models have helped researchers trace the evolutionary roots of the mechanism and understand its parallels in other vertebrates. In humans, though, the intricate architecture of these drains and veins appears uniquely adapted to balance protection and communication at an exceptional scale.
Economic and Healthcare Implications
While the immediate economic impact of this research may not be as tangible as in pharmaceuticals or technology startups, the long-term potential is immense. Understanding how skull veins adapt to immune demands could transform diagnostics and treatment approaches for a range of neurological conditions. Better predictive models could reduce hospital stays, improve outcomes, and optimize post-injury rehabilitation strategies.
This discovery also opens new opportunities for the biotechnology and imaging industries. Companies specializing in brain imaging, vascular modeling, and targeted immunotherapy may find value in developing products that visualize or manipulate these drainage systems. Improved understanding of skull-brain fluid dynamics could enhance resource allocation in neurotrauma care, potentially reducing national healthcare costs associated with chronic brain injury.
Regional comparisons show that research hubs in the United States, Europe, and East Asia are rapidly expanding their focus on neurovascular and immune connectivity. Stateside, institutions in California and Massachusetts are leading studies combining computational modeling with biological imaging to map the skull’s internal vasculature in greater detail. Asian research groups are focusing on genetic and molecular regulation aspects, while European teams emphasize aging and disease-related changes.
Challenges and Future Directions
Despite enthusiasm surrounding the discovery, significant questions remain. How do these venous border rearrangements occur at the molecular level? What signals trigger the veins to loosen or tighten their boundaries? And could these processes become dysregulated in disease?
Answering these questions will require interdisciplinary collaboration among immunologists, neurologists, and vascular biologists. Early evidence suggests that cytokines — small signaling proteins released during immune response — may play a role in initiating these vascular changes. Mechanical stress, blood flow dynamics, and metabolic signals could also factor into the process.
Advancements in live-imaging technologies and molecular tracing methods will allow scientists to watch immune cell migration through the skull in real time. Such insights may clarify how the body naturally controls inflammation in the brain and how modern medicine might learn to support that balance rather than override it.
A New Frontier in Brain Protection
The idea that skull veins can rearrange their borders to accommodate patrolling immune cells redefines one of biology’s oldest assumptions: that the brain is locked away behind an unyielding wall. Instead, it is guarded by a living, responsive system that actively manages its relationship with the immune world.
This subtle yet profound shift in understanding could influence not only medicine but also how humanity perceives its most vital organ. The skull, once seen merely as armor, now stands revealed as an adaptive frontier — one that protects the brain not through rigidity, but through intelligent, living flexibility.